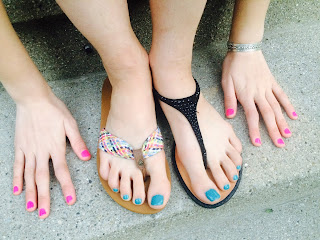What is supposed to be Susan's last clinical trial appointment is in a few hours.
I slept poorly last night, largely due, I am sure, to the new information that was swirling around in my mind.
The clinical trial coordinator called me late yesterday afternoon. She was calling to check to see how Susan had been tolerating her daily peanut dose since we started holding her Zyrtec (her last dose was early Thursday morning), and to see if we had any questions about the upcoming visit.
She was pleased (and pleasantly surprised, just a little, I think), to hear that Susan had been tolerating her dose well. (When I reported that, it was before Susan took her shower...and before the return of the light, hivey rash we have become familiar with over the last year.)
I took the opportunity to ask some questions about Susan's post-clinical trial care plan -- planning I am certain I have complicated in announcing our plan to seek private OIT treatment for Susan with Dr. Sakina Bajowala coupled with the request that one of the PRROTECT doctors co-follow Susan once she is discharged from the clinical trial.
As an aside, let me say that I think maybe, just maybe, this is where the idea of making meaning out of the senseless things that happen in this world comes in, for I know asking that a doctor from PRROTECT co-follow Susan with Dr. Bajowala could only be good...for Susan, for research, for anyone and everyone hoping for truly life-changing treatments for those living with food allergies. And yet, I also know that in asking for this, we are making a highly unusual request. I wrote a missive on this topic last Thursday to all of the doctors involved...and when (and if) appropriate, I will share it.
Basically, I explained why it was important to have our (peanut butter) cake and eat it, too.
In response to my missive, I learned that there is likely to be a 5-year follow-up study -- different from the one that did not get funded (Susan would not have qualified for that one, for all subjects had to achieve tolerance of 4000 mg of peanut). While it was really disappointing to us from a greater-good perspective that the original follow-up study was not funded, the one that has been proposed is likely to work out better for Susan.
There is far, far more that I do not know than what I do know.
And...
While at this point there are far, far more questions than answers, it seems like we just might...maybe, actually get to have our (peanut butter) cake and eat it, too.
While the protocol for the follow-up study is still being finalized, and they need IRB (Institutional Review Board) approval, the clinical trial coordinator was able to provide me with an overview.
In some ways, the follow-up study, which will consist of a single visit each year, will be significantly more flexible than the clinical trial. The clinical trial coordinator explained that in the follow-up study, there is likely to be more flexibility, allowing for the occasional skipped dose and fewer post-dosing activity restrictions. Since Susan's goal is to pursue greater tolerance of peanut, skipping of doses is irrelevant to us, and given that her tolerance of her daily dose is not reliable, I cannot see decreasing her activity restriction.
I did have one pressing question...the single issue that I knew might be a deal-breaker for Susan.
The question was this: "What is the plan in terms of dietary restrictions?" The clinical trial coordinator was not sure, but she was able to tell me that...at least as of right now...the discharge instructions remain the same as they have been during the course of the clinical trial: NO NEW FOODS.
I pressed her, knowing that while Susan has never been in this for the ice cream or bakery treat, she has (honestly, we all have) been looking forward to greater freedom when it comes to eating out.
The clinical trial coordinator acknowledged that one of the goals of the clinical trial is to afford subjects greater freedom when it comes to doing things like eating out, and trying new foods...but, she was unable to provide a clear answer on the question of dietary restrictions.
And so I asked more directly:
"Will subjects in the follow-up study be asked to adhere to the no new foods restriction that they have been following during PRROTECT?"
The clinical trial coordinator did not know, and while I appreciated her honesty, I was also stunned that this piece remains uncertain.
I had a few hours to process the information I learned during my conversation with the clinical trial coordinator before I saw Susan, who spent the day at the rink. I am glad I had some time to process what I had learned, for I was able to present what I did know clearly, and definitively...even though much of what I was presenting to Susan was, in fact, uncertain.
I hesitated when I got to the piece about the dietary restrictions...(knowing full well that I still had a group of Susan's friends cued up to surprise her at Baskin Robbins tonight, thinking that I should probably call the outing off...yet waiting, wanting to hear that these restrictions must remain in place directly from the doctor). I explained to Susan that, according to the clinical trial coordinator, at least as of right now (and for an indeterminate amount of time going forward), she is likely to have to observe the "no new foods restriction."
Susan took it all in, no questions.
And then she said, "I can do that. I want to do the follow-up study."
I was simultaneously thrilled, for I believe Susan's choice is right when it comes to the science of things...
And taken aback...
For what if she is asked to observe the "no new foods" restriction for the duration of the follow-up study? (I don't know if she will be, but I also do not know that she will not be...)
She will be a SENIOR in HIGH SCHOOL in five years.
Unfathomable, really -- to imagine the rest of Susan's childhood in a clinical trial follow-up...still living with her current dietary restrictions.
And so I asked some questions -- mostly wanting to be sure Susan understood what she was saying, and reminding her that, since the follow-up study protocol is not yet complete, she has some time to think about it (we will not be presented with consents today because they are not yet ready).
And yet, Susan remained clear. She wants to do the follow-up study. And she doesn't care what that means in terms of restrictions, "Because it is important, Mom."
And besides, she added..."It isn't any different than what we already do."
My heart catches and my breath hitches when I think about what continuing to follow the "no new foods" dietary restriction means...for Susan, for our family.
I feel conflicted (even after a night of tossing and turning) about Susan's clinical trial graduation gift -- a card tucked into an envelope with a host of $5 gift cards for ice cream and baked goods...and candies we have only ever looked at -- Snow Caps (my childhood favorite) and the new Hershey's chocolate covered caramels...
What do I do with that gift?
Give it?
Hold it?
Disassemble it, using the gift cards on Susan's siblings when she is not present?
A long night later, I still have not decided.
I am going to take Susan's gift with us when we head downtown to Ann & Robert H. Lurie Children's Hospital later today. I think I am going to leave it in the car...for I am not sure what I am going to do with it.
I am thankful I also tucked a bracelet into that gift bag...one with meaning, for no matter what happens, I can give her the bracelet.
Hours later, I haven't figured out how I feel about where we are now. (Well, some things I know...I am beyond thankful that there is likely to be a follow-up study...which I think is important for Susan, for the other children who are finishing PRROTECT at Ann & Robert H. Lurie Children's Hospital...and for the future of food allergy research and treatment.)
But more than anything...
This is where I step back and marvel at Susan.
For it is clear to me that, at least in her case, if she enrolls in the follow-up study, it will not be about Susan. Enrollment in the follow-up study will be all about the greater good.
And incredibly, Susan wants to participate in the follow-up study -- even with the uncertainties about dietary restriction...even if, as I put it, worst case, she has to follow the "no new foods" restriction for the next five years.
"Because it is important, Mom."
And so, on the morning of Susan's graduation from the clinical trial, I am struck by the fact that her future is as uncertain as many graduates' futures are on their graduation day. Uncertain as the future is however, I am certain Susan has grown from her experiences, and that she is in a better place now than she was eighteen months ago.
photo

Photo by Rebecca Gould Photography
Tuesday, June 30, 2015
Wednesday, June 24, 2015
Blogged Too Soon
I have always struggled with the idea that "everything happens for a reason" -- because what about those truly horrible things that defy reason? Children with cancer? Acts of terrorism...the unthinkable acts of violence my clients endured as children?
Then one day, a very wise man (who just happens to be married to my very dear, wise and insightful friend) said that (and I'm not quoting him, as I am sure he was more eloquent than I am about to be) it is up to us to make meaning out of the senseless things that happen in this world.
This idea resonated within me...and I have recalled his words often.
Today, when Susan and I set out for the city, I had her peanut dosing logs and her graduation card and gift with me. We also had a token of appreciation for the clinical trial staff and those who work in the Clinical Research Unit (CRU).
We dropped my car off for service, picked up a loaner and even though I pulled away from my mechanic's shop later than I had planned, we were making good time. Fears of being late slipped away, and I got lost in my own thoughts while Susan texted some of her early-riser friends. I noticed the northbound traffic (headed in the opposite direction) was barely moving, and I offered up a moment of thanks that that's not the direction we were going.
And then my phone rang -- and I recognized instantly that the caller was calling from Ann & Robert H. Lurie Children's Hospital. Hmmm. Since I was in a loaner car, my phone was not coupled with my car's hands-free system and in the seconds while I fumbled to answer the call, I wondered...finally deciding that it was a reminder call for one of Susan's post-operative follow-up appointments...
Except, it wasn't.
The minute I heard the clinical trial coordinator's voice, I knew our day wasn't going to be as planned. I think I heard the apology in her tone.
She explained -- she had forgotten to have us hold Susan's Zyrtec, and since the final day of the clinical trial required skin testing (which I either did not know or had completely forgotten), we had one of two choices -- go and do the physical examination, breathing tests, blood draw and return the following week for the skin testing or simply reschedule the entire appointment.
I explained the situation to Susan, and offered Susan the two options (thinking -- "control, give her control") and couldn't help but agree with her choice -- reschedule for next week.
At the next exit, I pulled off the highway, and over -- pausing as the world of early-morning commuters swirled around me. I checked my calendar and confirmed that we could be there the following week (for those keeping track, "Graduation Day" is now Tuesday, June 30, 2015).
I sent a brief text canceling a surprise I had planned for later in the day...
and I fought tears of disappointment.
I feel the loss keenly, and yet I determined (even before the reality of these losses crystalized in my mind) that if it was not "Graduation Day," I was going to make it a great day.
While I sat by the side of the road, I texted my good friend, Andi, who has manicured and pedicured Susan several times during the clinical trial -- especially last summer, when Susan did not feel well. I briefly explained...and asked if she had time for an "emergency" pedicure. She didn't -- but her friend did...
And I moved into damage control mode.
I proposed a pedicure, with book shopping before and sandal shopping after (I know, it would have been better to go sandal shopping before the pedicure, but I had to take the appointments available on short notice).
We were waiting when Half-Price Books opened...
And Susan did some serious damage to the clearance cart, where each of the four books she found were $1 or less...
This was Susan's second pedicure EVER, and she loved it!
AND...all day long she kept talking about how well the blue nail polish went with...just about everything (the shoes she was wearing, the pink polish on her fingers, the color of her bedroom)...I am so glad she is so pleased with it!
Susan's blue toenail polish does look nice -- with her pink fingernail polish, her new sandals AND the silver bracelets we have given her at key points in the clinical trial.
As we were driving toward the rink, Susan examined her freshly manicured toes and fingers and checked out her new shoes. We talked about the order in which she planned to read her new books...and Susan declared that we had filled the morning with "all of my happy and good things."
As the day wore on, the impact of Susan not graduating crystalized. I alternated between feeling a nearly crushing disappointment to reminding myself that one more week of living as we already do is...really, nearly nothing.
Except...for the losses I feel (perhaps) more keenly than Susan
I am eversothankful that Susan does not know I invited a group of her close friends to join us for ice cream at Baskin Robbins. She does not know that I scoped out the two different nearby Baskin Robbins locations and picked the one offering more flavors. She does not know that I made arrangements with the manager for her to try each and every flavor that does not contain nuts (she still has a tree nut allergy, and a mild soy allergy).
I am ever so thankful that Susan does not know that I planned for us to have dinner at Highwood Days, an event she loves that is like a giant community picnic, and a veritable "feast on foot." Each year, there are numerous vendors with interesting and unique food offerings, and Susan has only ever been able to eat pizza. I had envisioned a dinner on foot -- wandering with Susan, trying whatever looked good to her.
As the day wore on, my disappointment crystalized around the realities of what could no longer happen (at least not as planned).
I rescheduled Susan's Thursday-morning appointment to have her braces put on (we had agreed to wait until after the clinical trial to start orthodontic treatment, due to the increased risk of an open wound in the mouth, which some feel increases the risk of an adverse reaction during daily dosing).
I exchanged e-mail with the doctors overseeing the clinical trial about a road trip we intended to take this weekend. There is a significant stretch of our journey that is isolated, and our destination is remote. We revisited the wisdom of this trip, and at best, it will be...different than I envisioned. With the no-new-foods restriction, travel is exceedingly hard. I can (and will) pack food for Susan as I have always done -- I was just looking forward to greater freedom -- for her, and for me.
I contemplated our dinner reservations for Friday night (Susan's last night as an "only" before her twin siblings return home from camp), at what sounds like an incredible restaurant. We were planning to enjoy a "tasting menu" -- small portions of a number of unusual offerings. With the no-new-foods restriction still in place, I cannot see how this is still an option.
As the day wore on, something else happened, too, though -- we had the very beginning of what I hope will be important communication about Susan's treatment once she has completed the clinical trial. I have new hope that maybe, just maybe, there will be a partnership.
Intermingled with my disappointment is now hope -- and the feeling that maybe, just maybe, we can make meaning out of this disappointment.
Susan and I took a long walk with our puppies just before she went to bed.
We talked.
Susan shared her disappointment...
and we agreed that after having lived with the restrictions necessary to keep her safe given the severity of her peanut allergy for years, and the restrictions that came with the clinical trial for eighteen months, we can do this for one more week...
and we reflected on how fortunate we are that we have greater freedom to look forward to.
As the day wound down, I reflected on something my dear, wise friend wrote about Susan's clinical trial experience: "The definition of resiliency: embarking on a path, expecting one destination, ending up slightly off-course, and making a journey of it all." As difficult as the clinical trial has been at times, we have learned so much, and grown immeasurably, and I am thankful for every bit of our experience.
As I tucked Susan in, I gave her one more "happy and good thing" -- a miniature orchid.
Then one day, a very wise man (who just happens to be married to my very dear, wise and insightful friend) said that (and I'm not quoting him, as I am sure he was more eloquent than I am about to be) it is up to us to make meaning out of the senseless things that happen in this world.
This idea resonated within me...and I have recalled his words often.
Today, when Susan and I set out for the city, I had her peanut dosing logs and her graduation card and gift with me. We also had a token of appreciation for the clinical trial staff and those who work in the Clinical Research Unit (CRU).
We dropped my car off for service, picked up a loaner and even though I pulled away from my mechanic's shop later than I had planned, we were making good time. Fears of being late slipped away, and I got lost in my own thoughts while Susan texted some of her early-riser friends. I noticed the northbound traffic (headed in the opposite direction) was barely moving, and I offered up a moment of thanks that that's not the direction we were going.
And then my phone rang -- and I recognized instantly that the caller was calling from Ann & Robert H. Lurie Children's Hospital. Hmmm. Since I was in a loaner car, my phone was not coupled with my car's hands-free system and in the seconds while I fumbled to answer the call, I wondered...finally deciding that it was a reminder call for one of Susan's post-operative follow-up appointments...
Except, it wasn't.
The minute I heard the clinical trial coordinator's voice, I knew our day wasn't going to be as planned. I think I heard the apology in her tone.
She explained -- she had forgotten to have us hold Susan's Zyrtec, and since the final day of the clinical trial required skin testing (which I either did not know or had completely forgotten), we had one of two choices -- go and do the physical examination, breathing tests, blood draw and return the following week for the skin testing or simply reschedule the entire appointment.
I explained the situation to Susan, and offered Susan the two options (thinking -- "control, give her control") and couldn't help but agree with her choice -- reschedule for next week.
At the next exit, I pulled off the highway, and over -- pausing as the world of early-morning commuters swirled around me. I checked my calendar and confirmed that we could be there the following week (for those keeping track, "Graduation Day" is now Tuesday, June 30, 2015).
I sent a brief text canceling a surprise I had planned for later in the day...
and I fought tears of disappointment.
I feel the loss keenly, and yet I determined (even before the reality of these losses crystalized in my mind) that if it was not "Graduation Day," I was going to make it a great day.
And I moved into damage control mode.
I proposed a pedicure, with book shopping before and sandal shopping after (I know, it would have been better to go sandal shopping before the pedicure, but I had to take the appointments available on short notice).
We were waiting when Half-Price Books opened...
And Susan did some serious damage to the clearance cart, where each of the four books she found were $1 or less...
AND...all day long she kept talking about how well the blue nail polish went with...just about everything (the shoes she was wearing, the pink polish on her fingers, the color of her bedroom)...I am so glad she is so pleased with it!
Susan's blue toenail polish does look nice -- with her pink fingernail polish, her new sandals AND the silver bracelets we have given her at key points in the clinical trial.
As we were driving toward the rink, Susan examined her freshly manicured toes and fingers and checked out her new shoes. We talked about the order in which she planned to read her new books...and Susan declared that we had filled the morning with "all of my happy and good things."
As the day wore on, the impact of Susan not graduating crystalized. I alternated between feeling a nearly crushing disappointment to reminding myself that one more week of living as we already do is...really, nearly nothing.
Except...for the losses I feel (perhaps) more keenly than Susan
I am eversothankful that Susan does not know I invited a group of her close friends to join us for ice cream at Baskin Robbins. She does not know that I scoped out the two different nearby Baskin Robbins locations and picked the one offering more flavors. She does not know that I made arrangements with the manager for her to try each and every flavor that does not contain nuts (she still has a tree nut allergy, and a mild soy allergy).
I am ever so thankful that Susan does not know that I planned for us to have dinner at Highwood Days, an event she loves that is like a giant community picnic, and a veritable "feast on foot." Each year, there are numerous vendors with interesting and unique food offerings, and Susan has only ever been able to eat pizza. I had envisioned a dinner on foot -- wandering with Susan, trying whatever looked good to her.
As the day wore on, my disappointment crystalized around the realities of what could no longer happen (at least not as planned).
I rescheduled Susan's Thursday-morning appointment to have her braces put on (we had agreed to wait until after the clinical trial to start orthodontic treatment, due to the increased risk of an open wound in the mouth, which some feel increases the risk of an adverse reaction during daily dosing).
I exchanged e-mail with the doctors overseeing the clinical trial about a road trip we intended to take this weekend. There is a significant stretch of our journey that is isolated, and our destination is remote. We revisited the wisdom of this trip, and at best, it will be...different than I envisioned. With the no-new-foods restriction, travel is exceedingly hard. I can (and will) pack food for Susan as I have always done -- I was just looking forward to greater freedom -- for her, and for me.
I contemplated our dinner reservations for Friday night (Susan's last night as an "only" before her twin siblings return home from camp), at what sounds like an incredible restaurant. We were planning to enjoy a "tasting menu" -- small portions of a number of unusual offerings. With the no-new-foods restriction still in place, I cannot see how this is still an option.
As the day wore on, something else happened, too, though -- we had the very beginning of what I hope will be important communication about Susan's treatment once she has completed the clinical trial. I have new hope that maybe, just maybe, there will be a partnership.
Intermingled with my disappointment is now hope -- and the feeling that maybe, just maybe, we can make meaning out of this disappointment.
Susan and I took a long walk with our puppies just before she went to bed.
We talked.
Susan shared her disappointment...
and we agreed that after having lived with the restrictions necessary to keep her safe given the severity of her peanut allergy for years, and the restrictions that came with the clinical trial for eighteen months, we can do this for one more week...
and we reflected on how fortunate we are that we have greater freedom to look forward to.
As the day wound down, I reflected on something my dear, wise friend wrote about Susan's clinical trial experience: "The definition of resiliency: embarking on a path, expecting one destination, ending up slightly off-course, and making a journey of it all." As difficult as the clinical trial has been at times, we have learned so much, and grown immeasurably, and I am thankful for every bit of our experience.
As I tucked Susan in, I gave her one more "happy and good thing" -- a miniature orchid.
Tuesday, June 23, 2015
Graduation Day
It is early in the morning, and today -- clinical trial graduation day -- is a day we have looked forward to and talked about for...months.
Way back when...in the beginning of the clinical trial, before Susan had any real difficulties, we talked about "graduation day" with awe, reverence, hope...and optimism.
Way back when...I envisioned an enveloped filled with small gift cards -- to places Susan has never eaten -- The Bent Fork (a small, but incredible bakery near our house), Dairy Queen, Baskin Robbins, Frost, Culver's, Jolly Good Fellows Sweet Boutique, Dunkin' Donuts...and maybe some gift cards for interesting ethnic restaurants thrown in for good measure.
I think we thought it seemed like something truly incredible would happen between then and now.
And while something "truly incredible" has happened, our awe, reverence, hope and optimism have been tempered by the reality of the past eighteen months. While I tell myself that today should be even more meaningful because of the difficult road we have traveled, in reality, "graduation day" does not feel like the accomplishment I hoped it would...and I fear Susan feels the same way.
In the past two months, I have purchased nearly half a dozen graduation cards -- for my nephew's high school graduation, my good friend's son's high school graduation, a former sitter's graduate school graduation, and my sister-in-law's niece's college graduation.
Of course, I didn't go buy all of those cards all at once (that would have been too organized!), and so over the past two months, I have stood in the graduation card section of Target, Walgreens, CVS and Jewel.
Each time, I have contemplated purchasing a graduation card for Susan.
And each time, although I had Susan's upcoming graduation in the back of my mind as I browsed cards, I never saw anything that seemed "just right."
I have never wondered why I couldn't find a "just right" graduation card -- I am sure it is because of all of the mixed emotions that come with today's "graduation." While I know, and Susan knows -- and the doctors and clinical trial coordinators know -- and even most of those who have followed Susan's peanut eating journey know that while Susan never achieved the tolerance desired through the clinical trial, she has still accomplished something truly significant.
I spent a lot of time pondering the coincidence of the timing of Susan's graduation from the clinical trial and graduations in general before I made the resolve to purposefully shop for a graduation card for Susan.
It took three stops: CVS, Walgreens and then Target -- where eventually I found a card that felt "just right."
I have decided that even though we are not done with Susan's peanut eating journey, I am not going to let that take anything away from today. I am taking her card, filled with small gift cards...and a small gift bag with us to the clinical trial today.
I don't know if Susan will be surprised...but I think she will be.
We are going to celebrate Susan's clinical trial graduation day -- the commitment, the courage, the strength and the vision that it took to get here -- even though it is not quite what we hoped and dreamed and envisioned it might be.
Way back when...in the beginning of the clinical trial, before Susan had any real difficulties, we talked about "graduation day" with awe, reverence, hope...and optimism.
Way back when...I envisioned an enveloped filled with small gift cards -- to places Susan has never eaten -- The Bent Fork (a small, but incredible bakery near our house), Dairy Queen, Baskin Robbins, Frost, Culver's, Jolly Good Fellows Sweet Boutique, Dunkin' Donuts...and maybe some gift cards for interesting ethnic restaurants thrown in for good measure.
I think we thought it seemed like something truly incredible would happen between then and now.
And while something "truly incredible" has happened, our awe, reverence, hope and optimism have been tempered by the reality of the past eighteen months. While I tell myself that today should be even more meaningful because of the difficult road we have traveled, in reality, "graduation day" does not feel like the accomplishment I hoped it would...and I fear Susan feels the same way.
In the past two months, I have purchased nearly half a dozen graduation cards -- for my nephew's high school graduation, my good friend's son's high school graduation, a former sitter's graduate school graduation, and my sister-in-law's niece's college graduation.
Of course, I didn't go buy all of those cards all at once (that would have been too organized!), and so over the past two months, I have stood in the graduation card section of Target, Walgreens, CVS and Jewel.
Each time, I have contemplated purchasing a graduation card for Susan.
And each time, although I had Susan's upcoming graduation in the back of my mind as I browsed cards, I never saw anything that seemed "just right."
I have never wondered why I couldn't find a "just right" graduation card -- I am sure it is because of all of the mixed emotions that come with today's "graduation." While I know, and Susan knows -- and the doctors and clinical trial coordinators know -- and even most of those who have followed Susan's peanut eating journey know that while Susan never achieved the tolerance desired through the clinical trial, she has still accomplished something truly significant.
I spent a lot of time pondering the coincidence of the timing of Susan's graduation from the clinical trial and graduations in general before I made the resolve to purposefully shop for a graduation card for Susan.
It took three stops: CVS, Walgreens and then Target -- where eventually I found a card that felt "just right."
I have decided that even though we are not done with Susan's peanut eating journey, I am not going to let that take anything away from today. I am taking her card, filled with small gift cards...and a small gift bag with us to the clinical trial today.
I don't know if Susan will be surprised...but I think she will be.
Monday, June 22, 2015
The Plan...to Keep EATING PEANUT!
This is the e-mail I sent to the Principle Investigator and the other doctors and staff involved in the PRROTECT study at Lurie. I am sharing it because it lays out our thought process and next steps clearly, and I know many people who have followed Susan's journey are interested in knowing what our plan is. This morning, Susan joked that we will call this "Eating Peanut, Phase 2." And...perhaps we will.
Dr. P.,
Dr. P.,
As Susan’s completion date for the clinical trial draws near, I
wanted to write to thank you, Dr. R. and Dr. M – as well as (...) and (...) and the various other clinical trial coordinators we knew along the
way – for all you have done to support Susan during the clinical trial. While I know Susan did not achieve the level of
peanut tolerance anticipated, we are thankful beyond words that she achieved
the tolerance she did.
As you know, we have been very concerned about Susan’s occasional inability
to tolerate her daily dose of 2000 mg of peanut protein, as we wonder what that
means when it comes to protection against casual exposure and/or
cross-contamination. Susan’s seemingly
random anaphylactic reactions also leave us constantly just a bit on edge,
always slightly uncertain about how she will tolerate her daily dose. While I understand that with more than seven
months of daily dosing at 2000 mg of peanut protein Susan should be able to consistently tolerate her daily dose, as you
know, that has not been the case. Clearly,
Susan’s journey toward tolerance of peanut has been atypical, and we hope that
her commitment to the clinical trial proves beneficial to others.
Since our last clinical trial visit, I have done extensive
research and have spoken with a number of different people in the food allergy
community (researchers, doctors and parents).
As you know, despite (or maybe because of) how difficult the journey has
been, Susan is very clear that she
wants to continue eating peanut. While
she is open to the idea of decreasing her daily dose in the short term, in the long
term, she wants to be able to eat enough peanut daily that she does not have to
worry about miniscule variances in her daily dose causing anaphylaxis. While Susan appreciates the freedoms she is
likely to have as the result of the clinical trial (she has already, for
example, been to several movies in the theater and we are talking about taking
a short flight), she wants greater freedom.
I have become convinced that if Susan is to achieve long-term true
tolerance of peanut, we need to take a step back, to look at things
differently. I am interested, for
example, in looking at Susan’s gut health.
I am concerned that some of the very medications added early in the clinical
trial (to support her tolerance of peanut) might be interfering with her absorption
of peanut. I also believed the addition of
probiotics might make a difference in Susan’s ability to tolerate peanut. Recently, I have read some very interesting
things about the importance of Vitamin D in the diets of children with severe
food allergies. While I do not know if
the answer for Susan is as simple as any of the above ideas, I feel that we
must at least try things differently.
After extensive research and discussion, we have decided to work
with Dr. Sakina Bajowala, at Kaneland Allergy.
I understand that there are a lot of concerns about oral immunotherapy
offered in a private practice setting, and some of those concerns are what led
us to initially pursue treatment in a clinical trial for Susan. That said, as the clinical trial draws to a
close for Susan, we have become increasingly interested in pursuing a highly personalized
treatment for Susan. I have come to
believe that the best way to get a specialized treatment approach is in a
private practice setting with a doctor who has extensive experience in oral
immunotherapy.
We appreciated your approval of and Dr. R.'s willingness to
co-follow Susan with Dr. M. (Susan's primary allergist), and want you to know that our decision is
based on what we hope Dr. Bajowala can offer Susan. I remain hopeful that Susan’s journey – while
clearly outside the expected norm – is helpful for others, for surely there are
others like Susan. If you, or Dr.
R., would like to co-follow Susan during her post-clinical trial care and treatment
with Dr. Bajowala, she is amenable to that, and we would welcome the continuity
of care.
Thank you again – while our clinical trial experience was more
difficult than I ever could have anticipated, we now know the depth of Susan’s
strength and the strength of her commitment.
Thank you for giving her (and us) hope.
Caryn
Caryn
Sunday, June 21, 2015
Big, Big Decisions
It is a terrible, overwhelming feeling to realize that there are no good, clear treatment options for your child.
(And let me take a moment to say that when I realized this, I offered up a silent word of thanks that my child was not facing a life-threatening illness, although I am constantly aware of the ways in which Susan's food allergies threaten and limit her life. As overwhelming as this realization was, even at the time, I knew it could be worse.)
I have always known that Susan's peanut allergy was unusual in it's severity, and that is why I had such high hopes for the PRROTECT study. I really believed that Susan might truly benefit from oral immunotherapy for peanut supported by Xolair...
And she has.
And yet, as the end of the clinical trial has drawn near, it has become increasingly clear that while Susan benefitted from the use of Xolair to support her desensitization to peanut through oral immunotherapy, we have fallen far short of where I had hoped we might be.
Actually, Susan has fallen far short of the course outlined in the consent for the clinical trial, too. So, as much as I wish this were just about disappointment and a wish that Susan had gotten to a point where she could tolerate more peanut than she currently does, this is about something bigger -- this is about the question of what happens when a subject does not achieve what is expected during the course of a clinical trial. This is about the question of what could, what should -- what can safely -- happen once the clinical trial is over.
I have spent hours [actually, I am sure, if you add up all the minutes, all the moments in which Susan's clinical trial experience has crossed my mind, momentarily (or, honestly, longer) distracting me...and all the time I have spent reading research, e-mailing with people, talking to experts, doctors, other parents...the tally is more like days, maybe even weeks]...contemplating the future.
Nearly eighteen months ago, we sat in a room in the Clinical Research Unit (CRU) at Ann & Robert H. Lurie Children's Hospital with a clinical trial coordinator and reviewed the consent for the PRROTECT study. There was nothing new, as the clinical trial coordinator had provided us with a copy of the consent form prior to our initial visit. Susan and I went through the consent carefully, as the clinical trial coordinator asked us to review it again there, before signing it. I read. Susan read. Occasionally, we talked.
Mostly, though, we just read.
There was nothing new -- but there was something...different...about sitting there, reading the consent, knowing that if (when) I signed it, we would be embarking on the first step in what I hoped would be a journey toward greater freedom for Susan.
That something...different...pulled my mind this way and that.
I remember -- ever-so-casually -- asking about something I read...or rather, didn't read. The consent addressed the 8000 mg peanut challenge at week 31 (which later became a 4000 mg peanut challenge). What it did not say was what happened to those subjects who failed the challenge. And sitting there, in the serious space that the CRU is, I found myself thinking about that...turning what felt like an omission over in my mind. What did happen to those subjects who did not achieve tolerance of 8000 mg of peanut? The clinical trial coordinator was very reassuring -- explaining that really, every subject should reach "tolerance"...and that it was nothing to worry about.
I pressed on.
(Anyone who knows me well knows I always want to know...What if...?)
Well...What if...a subject doesn't achieve tolerance of 8000 mg of peanut protein?
I didn't like the answer, which was that if a subject failed the challenge (at the time it was 8000 mg of peanut protein, but by the time Susan reached Week 31 of the PRROTECT study, the challenge had been reduced to 4000 mg of peanut protein), the subject would not be allowed to continue eating peanut once the clinical trial concluded.
Oh.
Well.
I allowed myself to be assured -- this kind of outcome was not the expectation.
Perhaps in an exceptional case, the subject would not achieve the desired tolerance, but as the PRROTECT study was specifically designed for children with extremely severe peanut allergies (just like Susan), I allowed myself to believe that Susan would not be one of "those subjects."
I heard the clinical trial coordinator explain that "those subjects" would not be allowed to continue consuming peanut at the conclusion of the clinical trial.
I heard that...and I thought...even if Susan was one of "those subjects"...surely some tolerance would be better than none. This idea flitted across my mind...unbidden and unwelcome...and I remember pushing it away, choosing to focus on the likelihood of success.
This idea of a "failure" flitted across my mind...unbidden and unwelcome...and I remember pushing it away, choosing to focus on the likelihood of success.
MindoverMatter.
The Power of Positive Thinking.
(And all that).
But now that Susan IS "one of those subjects," not a "subject" but a child on the cusp of adolescence (becoming her own person), the reality is stark, and sadly underscores one of many problems with clinical trials involving real people.
For while there is no protocol that allows Susan to continue eating peanut, how could anyone really ask -- or even expect -- Susan to stop eating peanut at this point? And even if -- within the protocol and the guidelines of the clinical trial -- there is no space for Susan to continue eating peanut, how could we -- as her parents -- support anything other than Susan's continued consumption of peanut?
We have -- in the last 18 months -- seen Susan safely go to the movies...not once, not twice, but countless times. We have even allowed her to go to the movies without an adult present. Susan has started thinking about those ethnic foods she wants to try (the list is long -- with Thai, Indian and Greek topping the list), about flying (Greece is high on her list -- but I'm in for an over-ground-only flight only first!) and about ice cream, donuts...and baked goods. I said it once, and I will say it before -- Susan is a foodie trapped in a food-allergic child's body.
I have heard that some "subjects" have not wanted to continue eating peanut.
But, Susan, is not a "subject" -- she is my daughter.
And she very much wants to continue eating peanut.
Despite the difficulties Susan has had (or maybe because of them), Susan has never expressed a desire to stop eating peanut. She has never (even when she was eating the Nutter Butters she found "disgusting") complained about taking her daily dose.
How could we, as Susan's parents, support anything other than Susan's continued Eating (of) Peanut?
While I would much rather take the road more traveled when it comes to anything medical, I started examining roads less traveled. And while the idea of a road less traveled alarmed me initially, I found myself warming to the idea...
1) STOP eating peanut. (Unimaginable to Susan, and, frankly, given what she has gone through, I did not see how we could ever, as parents, take a stance that required her to give up her hard-earned daily peanut dose, despite the risks it seems to carry.)
2) STAY the course. Keep at it. Hope that, with time, Susan's body will come to more "gracefully" accept (read: tolerate) her daily peanut doses. While we never actually hammered out the details, with discussion, it became apparent that the doctors involved in the clinical trial would likely support Susan in continuing to eat some amount of peanut on a daily basis. However, simply keeping at what we have been doing felt a bit like insanity. If one thing has become clear, it is that Susan does not easily tolerate her daily 2000 mg dose of peanuts. She has had several epinephrine requiring reactions to her daily dose over the last eight months, and a handful of concerning incidents (where we did not administer epinephrine, but watched ever so closely) -- the single episode of vomiting, the hives, the flushed cheeks and chest...
3) TRY SOMETHING NEW. Keep eating peanut, but re-examine things. I have long felt that Susan barely tolerates (at times) her daily 2000 mg dose of peanut protein. The random hives and flushed cheeks and single, seemingly random episodes of vomiting are, to me, constant reminders of the edge of the precipice upon which we walk. I know the researchers believe that this far in, a minute variance in dose should not make a difference in Susan's tolerance of her daily dose, but I also know that Susan's experience is different. For some reason that is not year clear to us, Susan does not reliably tolerate her daily dose. As I gathered information and assessed options, I found myself wondering about the medications Susan takes daily (Famotidine, Omeprazole and Zyrtec), and wishing we could add probiotics and maybe even Vitamin D.
In the weeks that followed Susan's second-to-last clinical trial visit, I reached out to many people -- doctors, researchers, pediatric allergists, parents of children living with food allergies, and friends who have followed our journey from the beginning. I did more reading than I did before signing the consent for Susan's participation in the clinical trial (and I did A LOT way back then), and a plan started to form.
I am beyond thankful to those who gave of their time, who answered my questions, who helped me think things through. I especially appreciate those who wear multiple hats -- parents of children with food allergies who are also researchers or allergists...and parents of children with food allergies who are also advocates of research. Everyone agrees that there is no easy answer. Everyone supports Susan's desire to keep eating peanut. One dear, wise friend observed how fortunate we are that we have options, and the wherewithal to pursue them -- even if none of the options seemed quite perfect.
In the end, we have what feels like a great next step. We are in unchartered territory -- few "subjects" find themselves in the position Susan will be in at the end of the clinical trial -- with no clear "next step." While I am sorry that Susan did not achieve the outcome desired in the clinical trial, I am thankful beyond words for the doctors and researchers who have gotten Susan to this point. We are thankful for all that they did to get Susan to the tolerance of peanut she has achieved, and we are immeasurably optimistic about the future...
Subscribe to:
Comments (Atom)